Hemostasis and Inflammation
Sepsis and Acute Kidney Injury
Sepsis is the tenth leading cause of death in the US with an estimated 750,000 cases per year. Sepsis is a life-threatening systemic inflammatory disease initiated by toxins associated with a bacterial infection which can lead to hypotension, organ failure, and death. The hemostasis system is a well-established target for bacterial toxins, and alterations in the activation of procoagulant and fibrinolytic systems together with strong proinflammatory responses are thought to play significant roles in the development of sepsis.
Sepsis initiated by gram-negative bacteria is often accompanied by acute kidney injury (AKI). Development of AKI during sepsis is associated with a higher incidence of mortality, increased morbidity, and often results in multiple organ damage. Currently, no effective treatment is available for preventing life-threatening septic AKI, due to a general lack of mechanistic relationships between the inflammatory response, inflammatory signaling pathways, and end-organ failure. Therefore, clearer understanding of the cellular and molecular mechanisms of the processes involved in the development of septic AKI will facilitate in uncovering new therapeutic approaches.
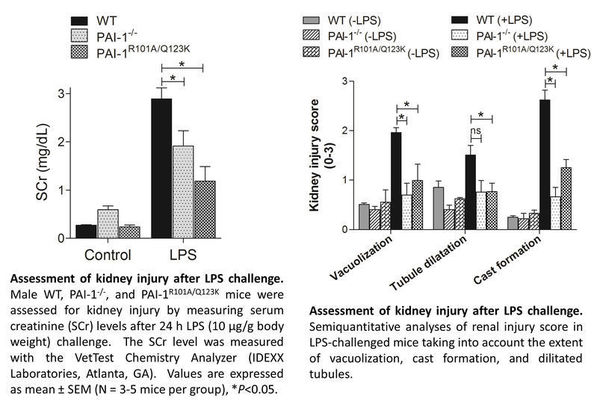
The goal of the W.M. Keck Center's current study is to determine the role of components of hemostasis in the progression of septic AKI by utilizing in vivo, in vitro and cell-based techniques. Researchers are currently focusing on the plasminogen activator inhibitor-1 (PAI-1), because elevated levels of PAI-1 have been observed in patients with severe sepsis, and are commonly associated with unfavorable outcomes and increased mortality, while the reduction of PAI-1 levels is associated with improved survival in patients. The team is utilizing a LPS-induced model of endotoxemia in wild-type (WT), PAI-1 deficient (PAI-1-/-) mice, and mice expressing a mutant PAI-1 with significantly diminished vitronectin binding capacity (PAI-1R101A/Q123K) in order to gain insights into the role of PAI-1 in response to septic AKI.